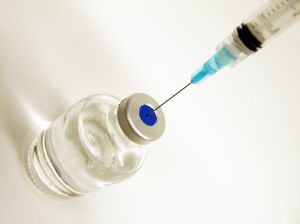
 The New England Compounding Center, a pharmacy located in Framingham, Massachusetts that distributed a steroid linked to an outbreak of fungal meningitis has issued a voluntary recall of all of its products.
The New England Compounding Center, a pharmacy located in Framingham, Massachusetts that distributed a steroid linked to an outbreak of fungal meningitis has issued a voluntary recall of all of its products.
The NECC announced the recall Saturday, claiming they were doing so because of the potential risk of contamination. As of now, they have no indication that any of their other products have been tainted.
The Food and Drug Administration has warned all health professionals not to use any of the products distributed by the center.
According to the Centers for Disease Control and Prevention, as of Sunday night, there have been 91 confirmed cases of fungal meningitis. This rare outbreak has affected nine states and left seven people dead.
The states with reported cases of fungal meningitis include Florida, Maryland, Indiana, North Carolina, Ohio, Tennessee, Michigan, Minnesota and Virginia.
Health officials are working to notify anyone who may have received an injection. At this time, it is unknown exactly how many people have been affected, though it could involve hundreds even thousands of people who received the steroid injections for back pain from July to September of this year.
The NECC is apparently fully cooperating with investigators.
While fungal meningitis is not contagious like its more common bacterial and viral counterparts, it can cause serious injuries and even death.
According to the CDC, those infected suffered from the following symptoms approximately 1 to 4 weeks after their injection: nausea, headache, fever and a neurological deficit consistent with deep brain stroke. Health officials claim that the steroids may have been contaminated by a dangerous fungus found in leaf mold. In the state of Florida, at least four meningitis outbreaks have been confirmed by the CDC.
The following products distributed by the NECC have been recalled:
Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #05212012
Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #06292012
Methylprednisolone Acetate (PF) 80 mg/ml Injection, Lot #08102012
Possibly contaminated products were received by the following facilities in Ocala, Florida:
FLORIDA PAIN CLINIC – 3241 Southwest 34th Street Ocala, FL 34474
MARION PAIN MANAGEMENT CENTER – 1737 Southeast 28th Loop Ocala, FL 34471
SURGERY CENTER OF OCALA – 3241 Southwest 34th Street Ocala, FL 34474
Can You File a Lawsuit?
If you or someone you care for has received an epidural steroid injection in the last three months and suffered a stroke or developed fungal meningitis, you may be able to obtain compensation for your pain and suffering. The Ocala Meningitis Outbreak Attorneys at Whittel & Melton can help gather the facts surrounding your injuries and further investigate whether or not the use of a specific medical product may have contributed to your suffering. We work with medical professionals and health care experts to establish the most up-to-date facts and relevant evidence to support your case.
Under state and federal laws, manufacturers and distributers of dangerous or defective products, can be held responsible for any injuries or harm their products cause. Manufacturers of medical products must make sure their products are safe to use before distributing them for public use. When they fail to do so, this can be considered negligent and a personal injury or wrongful death lawsuit can be filed on behalf of the victim.
These cases can be complex, and it is important to realize that there are statutes of limitations placed on how long you have to file suit. If you are unsure of whether you have a valid reason to file a claim, contact the Ocala Meningitis Outbreak Lawyers at Whittel & Melton online or call us in Marion County at 1-352-369-5334. We provide free and confidential consultations and are able to make house calls and hospital visits. Don’t delay, contact us today to learn about the legal options available to you.
 Florida Personal Injury Lawyers Blog
Florida Personal Injury Lawyers Blog